|
• Objective: To determine the variability
in the size and distribution of the first septal perforating-artery (FSPA).
« Material and Methods: In this pilot study, 10 fresh autopsy hearts
from patients who did not have hypertrophic cardioniyopathy (HCM) or
clinical evidence of coronary artery disease were evaluated for the
variability in the size of the FSPA. The size of the FSPA was also
measured during coronary angiography in 8 patients with HCM who were
undergoing alcohol septal ablation.
• Results: Of the 10 autopsy hearts, 2 had a large FSPA (>1.0 mm in
maximal diameter) with prominent septal myocardial distribution, 2 had a
medium-sized FSPA (0.5-0.9 mm), 2 had a small FSPA (0.1-0.4 mm), 3 had a
tiny FSPA (<0.1 mm), and I had an indiscernible ostium. In 2 patients
the FSPA supplied (he right ventricular free wall. In 4 patients the
basal ventricular septum was incompletely supplied by the FSPA. Of the 8
patients with HCM, the FSPA was larger than 2 mm in diameter in 2
patients, 1 to 2 mm in 4, and smaller than 1 mm in 2. The distance
between the left anterior descending coronary artery ostiuni and the
origin of the FSPA ranged between 13.1 and 37.4 mm, indicating a large
variation in the size and distribution nl the FSPA.
• Conclusions: Variability in the size and distribution of the FSPA in
patients without HCM was substantial. Areas of the heart other than the
basal septum were supplied in some patients by the FSPA. In other
patients the FSPA did not supply the entire basal septum. Similar
findings were noted in patients with HCM. A detailed evaluation uf the
distribution of the FSPA may be necessary in all patients with HCM who
are undergoing alcohol septal ablation.
Patients with hypertrophic cardiomyopathy (HCM) can present with
disabling symptoms of dyspnea, angina, or exertional syncope due in part
to obstruction of the left ventricular outflow tract.1-3 Despite optimal
medical therapy, some patients have persistent severe symptoms caused by
outflow tract obstruction. Septal myeclomy has been considered the
treatment of choice in these patients.4-6 As an alternative,
dual-chamber pacing has been advocated, but only a small percentage of
patients have a modest reduction in gradient and sustained symptomatic
improvement.7-9
Recently, catheter-based infarction of the ventricular septum supplied
by the first septal perforating artery (FSPA) of the left anterior
descending coronary artery was proposed as a nonsurgical method to treat
patients with severely symptomatic HCM.10-13 Because the FSPA suplies
the portion of the ventricular septum that contributes to subaortic
stenosis in most patients with the obstructive form of MCM, knowledge of
the size and distribution of this artery is important. The purpose of
this pilot study was to determine the variability in the size and
distribution of the FSPA. Initially, the distribution of the FSPA was
studied in 10 fresh autopsy hearts with use of an injection of barium
sulfate and gelatin. Subsequenlly, the distribution of the FSPA was
evaluated in 8 patients with HCM who were undergoing alcohol septal
ablation.
MATERIAL AND METHODS
We studied 10 fresh autopsy hearts from patients who did not have HCM or
clinical evidence of coronary artery disease. This study was approved by
the Mayo Foundation Institutional Review Board. The ascending aorta was
transected, and the left main coronary ostium was identified. Next, the
left main and left anterior descending coronary arteries were opened
longitudinally from above. The origin of the FSPA was the first
discernible ostium in the left anterior descending coronary artery along
its septal aspect. A solution of diluted barium sulfate and gelatin was
manually injected into the ostium of the FSPA through a 26-gauge needle
attached to a 10-mL syringe.14 Radiographs were taken in a right
anterior oblique projection to delineate the size and distribution of
the FSPA in the intact heart. Hearts were fixed in formalin for 24
hours. Short-axis ventricular sections, 1 cm thick, were. obtained, and
radiographs of all slices were prepared to show the distribution of the
FSPA.
The distribution of the FSPA was then evaluated clinically in 8
consecutive patients with HCM who were undergoing alcohol septal
ablation. In each of these patients, selective coronary angiography was
performed by using a right anterior oblique view with cranial angulation
to delineate the FSPA. A quantitative computer analysis program with
automatic border detection was used to measure the following: (1) the
mean diameter of the left anterior descending coronary artery proximal
to the FSPA, (2) the distance from the ostium of the left anterior
descending coronary artery to the FSPA, (3) the maximal diameter of the
FSPA, (4) the distance from the FSPA to the second septal perforating
artery, and (5) the maximal diameter of the second septal perforating
artery.
Table 1, Variability in the Size and Distribution of the First Septal
Perforating Artery
in 10 Autopsy Hearts*
*FSPA = first septal perforating artery; NA = not available.
RESULTS
Of the 10 autopsy hearts, 2 had a large FSPA (>1.0 mm in maximal
diameter) with prominent seplal myocardial distribution, 2 had a
medium-sized FSPA (0.5-0.9 mm), 2 had a small FSPA (0.1 - 0.4 mm), 3 had
a tiny FSPA (<0.1 mm), and 1 had an indiscernible ostium (Table 1,
Figures I and 2). In the 7 hearts with arterial measurements, the
diameter of the FSPA at its origin ranged from 0.05 to 1.8 mm (mean, 0.6
mm). Based on body weight and heart weight, FSPA diameters ranged from
0.0005 to 0.020 mm/g (mean, 0.009 mm/g) and 0.00012 to 0.004 mm/g (mean,
0.0016 mm/g), respectively.
In 2 patients, the FSPA also supplied the anterior wall of the right
ventricle. In the 4 patients with an FSPA less than 0,1 mm in diameter,
the basal septum was incompletely supplied by the FSPA.
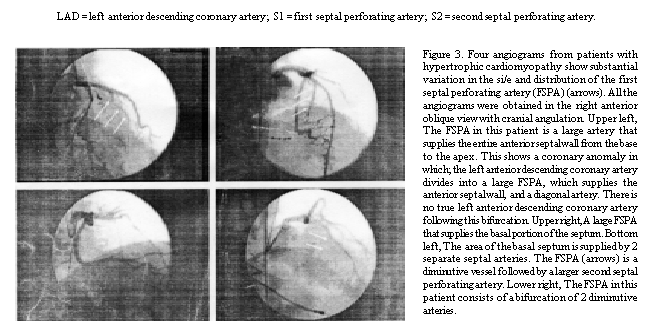
Of the 8 patients with HCM, 2 palienis had an FSPA larger than 2.0 mm in
diameter, 4 had an FSPA between 1.0 and 2.0 mm in diameter, and 2 had an
FSPA smaller than 1.0 mm in diameter. In 2 patients, the first and the
second seplal perforating arteries arose from a common ostium. The
distance between the ostium of the left anterior descending coronary
artery and the origin of the second perforating artery varied between
13.1 and 37.4 mm, indicating a large variation in the length of the
septum supplied by the FSPA (Table 2, Figure 3).
DISCUSSION
The pathophysiology of HCM is complex and includes abnormalities of
rhythm, diastolic function, and systolic function.2,3 In a subset of
patients, symptoms are due to a dynamic left ventricular outflow tract
obstruction from a hypertrophic septum and systolic anterior motion of
the mitral valve. Therapy for patients with severe obstruction has been
directed at reducing the obstruction of the dynamic left ventricular
outflow tract. 2,4,7 Surgical myotomy or myectomy alleviates symptoms in
more than 90% of patients.4-6
In most people the FSPA supplies the basal portion of the ventricular
septum. Investigators have reported the use of catheter-based
alcohol-induced thrombosis of the FSPA 111 the treatment of the patients
with HCM.10-13 Sigwart10 found that transient occlusion of the FSPA
caused akinesis of the basal ventricular septum and reduction in the
outflow gradient. With use of a catheter-based technique, alcohol was
instilled into the FSPA, creating an arterial occlusion and an infarct
of the basal ventricular septum. This resulted in a lowering of the
degree of outflow tract obstruction and a reduction in symptoms. A
postinfarction remodeling effect also occurs to the point that the size
of the outflow tract increases with time after successful ablation.
Although an increasing number of patients are rcceiving catheter-based
septal ablalion, the long-term outcome is unknown, and the ultimate role
of this procedure is not defined.10-12,15,16 Our findings indicate that
the FSPA varies widely in size and distribution from patient to patient.
It is important to understand the distribution and variation of the FSPA
when septal ablation is being performed. Some patients had an FSPA that
perfused only a smoll portion basal septum, whereas in others it not
only perfused the septum but also perfused the entire right ventricular
wall. Thus, occlusion of the FSPA may have a minimal effect in one
patient but may cause transmural infarction, right ventricular
infarction, or septal rupture in another patienl. This wide variation in
the size and takeoff was also seen in patients with HCM. Our findings
are consistent with the clinical observation that other seplal
perforators may be necessary for ablation, depending on the size of the
FSPA and the site of obstruction.
The current pilot study found a substantial variability in the size,
distribution, and perfusion regions of the FSPA in patients without HCM.
A large variation in the size and takeoff of the FSPA was also
identified in patients with HCM. These findings indicate that successful
septal ablation may not be possible in all patients if only the FSPA is
injected. Also, alcohol injection into the FSPA has the potential to
create large infarctions of the septum and right ventricle. Meticulous
evaluation of the distribution and perfusion of the septal arteries
before ablation may be necessary for optimal patient care.
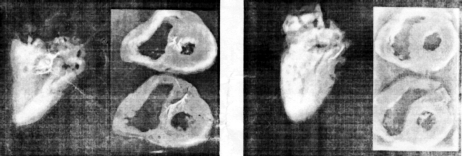
Figure 1. Postmortem radiographs of a
heart with a large first septai perforating artery. Left, Intact heart
has a prominent branching artery (right anterior oblique view). Right,
Ventricular slices show extensive distribution within the basal septum
(short-axis view).
Figure 2. Postmortem radiographs of a heart with a diminutive first
septal perforating artery. Left, Intact heart has a small arterial twig
(right anterior oblique view). Right, Ventricular slices show minimal
distribution within the basal septum (short-axis view).

 |
Literature:
1. Louie EK. Edwards LC 111.
Hypertrophic cardiomyopathy. Prog Cardiovusc D/.s-. 1994:36:275-308.
2. Maron B.I. t lyperlrophic curdiomyopathy. Curr Prohl Cunliul. 1993;
18:639-704.
3. Wigle EU, Sasson Z. Henderson MA, et al. Hypertrophic cardiomyopathy:
the importance of the site and the extent ot'hypertrophy: 11 • a review.
Prog Cunlioviisc Dis. 1985;28:1-83.
4. Mclntosh CL, Maron B.I. Current operative treatment of obstructive
hypertrophic cardiomvopathv. Circulation. 1988:78:487-495.
5. Mohr R. Schaff HV, Puga F.1, Danielson GK. Results of operation for
hypenrophic obstructive cardiomyopathy in children and adults less than
40 years of age. Circulation. \ 989,80(3, pt 1): 1191 - 13. 1196.
6. Mohr R, Schaff HV, Danielson GK, Puga FJ, Pluth JR, Tajik AJ. The
outcome of surgical treatment of hypertrophic obstructive cardiomyopathy:
experience over 15 years../ Thoruc Cunlioviisr Surg. 1989;97:666-674.
7. Fananapazir L, Cannon RO 111, Tripodi D, Panza JA. Impact of 15.
dual-chamber permanent pacing in patients with obstructive hypertrophic
cardiomyopathy with symptoms refractory to vcrapiimi' and
beta-adrenergic blocker therapy. Circulation. 1992,85:2149-2161.
8. Maron BJ, Nishimiu-a RA. McKenna W.1, Rakowski H, Josephson 16. ME,
Kieval RS. Assessment t)f permanent dual-chamber pacing as a treatment
for drug-refractory symptomatic patients with obstructive hypertrophic
cardiomyopathy: a randomized, double-blind, crossover study (M-PATHY).
Circulation. 1999,99:2927-2933.
9. McDonald K. McWilliams K, O'Keefe B. Maurer B. Functional assessment
of patients treated with permanent dual chamber pacing as a primary
treatment for hypertrophic cardiomyopathy. Eiir Hrui i J.
1988,9:893-898.
10. Sigwart U. Non-surgical myocardial reduction for hypertrophic
obstructive cardiomyopathy. Lancci. 1995.346:211-214.
11. Seggewiss H. Gleichmann U, Faber L, Fassbender I), Sclimidt UK.
Strick S. Percutaneous transluminal septal inyoctirdi;il .ihlation in
hypertrophic obstructive cardiomyopathy: acute results and 3-montii
follow-up in 25 patients../ Am Coil Cardiol. 1998:3 1:252-25S.
12. Knight C. Kurbaan AS, Seggewiss H, et al. Nonsurgical septiil
reduction for hypertrophic obstructive cardiomyoputhy: outcome in the
first series of patients. Cirriiltilion. 1997,95:2075-2081.
13. Lakkis NM, Nagueh SF, Kleiman NS, et al. Echocardiography-guided
ethanol septal reduction for hypertrophic obstructive cardiomyopathy.
Circulation. 1998,98:1750-1755.
14. Lie JT. Heart and vascular system. In: Ludwig .1. yd. Current
Methoiis of Autopsy Practice. 2nd ed. Philadelphia, Pa: WU SaundersCo;
1979:21-50.
15. Aguilar OM, Meyer D, Fromm RE, Spencer WH 111. Relationship of
number ofseplal branches injected and creaiinine kinase area under the
curve in nonsurgical septal reduction therapy for hyper-trophic
obstructive cardiomyopathy [;ihstraci|../ Am Coil Curdiiil.
2000;35(suppl A):88A.
16. Seggewiss H, Faber L, Meissner A. Meyners W. Kniter L. Zeimssen P.
Improvement of acute results after percutaneous transluminal septal
myocardial ablation in hypertrophic cardiomyopathy during mid-term
follow-up [abstract]. J Am Coil Cardiol. 2000:35(suppl A):188A. |


