|
Abstract
BACKGROUND: Tromboembolism is a feared complication following heart
valve replacement. Cerebral microembolic signals may be detected using
Doppler ultrasonography. Doppler ultrasound has been used to detect
microemboli during CPB. MES has also been detected in association with
myocardial infarction, left ventricle aneurysm, atrial fibrillation and
carotid artery stenosis. The aim of the present study was to examine the
frequency of MES in patients one year after heart valve replacement, to
look for possible risk factors associated with MES and for any
correlation with cerebral events.
MATERIALS AND METHODS: One hundred patients (mean age was 66,3
(+\-12,4), 69 male and 31 female) were examined one year after heart
valve replacement (group A). Thirty patients who had undergone various
cardiovascular operations but without heart valve pathology (mean age
was 62,5 (+\- 8,7), 39% male and 61% female) served as controls (group
B). A newly developed microemboli detector, EMEX-25 (Hatteland
Instrumentering, Norway) was used to detect microembolic signals from
both carotis arteries.
RESULTS: In group A MES were detected in 62 %, in group B in 46% of the
patients. The difference between valve patients and non-valve patients
was not statistically significant (p=0.2). In group A a correlation was
found between the number of MES and postoperative stroke, smoking,
previous cardiovascular operations and the EUROScore (p<0.05). There was
no correlation between the total number of MES and anticoagulation (INR
and anticoagulation therapy), atrial fibrillation, cholesterol level,
NYHA class, gender, age, valve type or valve position. In group B a
correlation was found for age, elevated serum creatinine level (>200
uMol/L), atrial fibrillation and EUROScore. Cerebral events were
diagnosed in 15 patients in group A and their correlation with the total
number of MES was statistically significant.
CONCLUSION: MES were detected in valve patients as well as in non-valve
patients one year after surgery. The difference between the two groups
was not statistically significant. The association between possible risk
factors and MES varied between valve patient and non-valve patients. A
strong correlation between number of MES and postoperative cerebral
events was found.
1. Introduction
Tromboembolism is a feared complication following heart valve
replacement. Despite adequate anticoagulation, the frequency of
tromboembolism ranges between 1-4 % per patient-year(1). Although
persistent cerebral injury occurs in only around 3% of patients,
cognitive impairment may occur in 2/3 of patients early after surgery
and persist in 1/3 of patients for at least one year(2).
Cerebral microembolic signals may be detected using Doppler
ultrasonography (3).
Experimental studies have shown that emboli passing a vessel have some
typical characteristics with regard to duration, direction and intensity
of the Doppler signal(4)
Doppler ultrasound has been used to detect microemboli during CPB(5,6).
Air bubbles have been detected in patients undergoing cardiac
surgery(7-10). In 1990 Spencer et al. monitored carotid endarterectomies
and detected Doppler signals, similar to those reflected from gas
bubbles, during arterial dissection, before opening the vessel. They
suggested that these signals were due to formed elements. In 1991 it was
confirmed that Doppler ultrasound could be used to detect solid as well
as gaseous microemboli by introducing microemboli of different sizes and
compositions in animal studies(11). Most studies in patients undergoing
open heart surgery have been performed using transcranial Doppler
ultrasonography(12-17) .
Microembolic signals have been detected during surgery in patients
undergoing valve replacement 1818 as well as in patients undergoing
CABG(18-21).
A significant association between intraoperative microembolic signals
and postoperative neuropsychological deficit has been
demonstrated(22,23). However, no association between neuropsychological
deficit and total number of MES could be demonstrated one year after
open heart surgery(22). MES have also been detected in association with
myocardial infarction, left ventricle aneurysm, atrial fibrillation and
carotid artery stenosis in patients (24-27).
The present methods have not been able to separate between solid and
gaseous emboli. However, the lack of correlation between the level of
anticoagulation and MES in one study may indicate a gaseous nature of
the emboli. Also several experimental studies attempted to confirm the
gaseous nature of MES produced by prosthetic valves (30-32) .
We have chosen the carotid artery for the Doppler study, where the
highest number of MES can be detected(33), and because of the simplicity
of the method.
The aim of the present study was to examine the frequency of MES in
patients one year after heart valve replacement, to look for possible
risk factors associated with MES and for any correlation with cerebral
events.
2. Materials and methods.
Patients
Group A included 100 patients one year after heart valve replacement. A
total of 72 patients had undergone AVR, 19 MVR and 9 DVR. In 63 patients
concomitant procedures had been performed, of which CABG was the most
frequent (39 patients). The most frequently used valves were
Carbomedicsâ (53 patients) and On-Xâ (40 patients), the remaining cases
received bioprostheses.
All patients were on anticoagulation therapy, aiming at INR levels of
2.5-3.5. The anticoagulation level (INR) was measured on the day of
follow-up.
The mean age for group A was 66.3 ( +12.4), 69 were male and 31 were
female.
Thirty age and sex-matched patients who had undergone major surgery
except valve replacement served as controls (group B). In these patients
CABG and various types of operation on the thoracic aorta had been
performed.
The mean age for group B was 62.5 ( + 8.7), 39% were male and 61% were
female.
The EUROScore was calculated for all patients. This score includes
information about age, gender, previous operations, accompanied diseases
and severity of the surgery (34). The two groups were comparable with
regard to age, gender, valve position, valve size and the most common
risk factors (smoking, reoperations, atrial fibrillation, arterial
hypertension, different hematological data and accompanied diseases).
Technical methods:
A newly developed microemboli detector, EMEX-25 (Hatteland
Instrumentering, Norway) was used for the measurements. The instrument
was connected to a PC running the EMMON.exe software program for signal
processing and for deriving of microemboli statistics.
A non-focused probed with the diameter of ten mm. was applied lateral to
the trachea with the measuring point at the “root” of the common carotid
artery. The probe was orientated towards the flow direction with an
angle relative to the skin of approximately 45 degrees. The sampling
depth was set to 2-3 cm.
 |
|
 |
|
Picture 1: EMEX-25 (Hatteland Instrumentering, Norway)
Picture 2: Measurement procedure
Coherent ultrasonic pulses with a resonance frequency of 3 MHz were used
and the Doppler shift of the becscattered signal was derived. The
instrument was optimised for the purpose of detection amplitude pulses
of the Doppler signal (originating from microembolies) with the highest
possible sensitivity. No discrimination between ultrasonic microbubbles
and solid particles was made.
Doppler signal amplitudes obtained from normal blood flow exhibited a
predictable behaviour with respect to variance of signal amplitudes.
However, when microembolic events occurred, the signal energy would
suddenly increase significantly when the microemboli was passing the
sample point of the ultrasonic probe. These amplitude bursts, termed MES
(Micro Emboli Signals), sounded like short chirps or clicks and were
easy to identify after some training.
EMEX-25 derived the envelop of the Doppler amplitude signal and assessed
a reference level which was twice (6 dB) in amplitude compared to the
average of the envelope signal. Any signal burst with an envelope
amplitude crossing this reference value was regarded to be a potential
microemboli. An artefact removal algorithm was applied to the spikes.
The algorithm basically analysed the curvature of the mean velocity to
evaluate if the spike was more susceptible to be caused by an artefact
or not.
The EMEX-25 was tested in 25 healthy volunteers. No MES were detected
examining both carotids for two minutes each.
Statistical analysis:
The chi-square test or Fisher’s exact test, whenever appropriate, were
used to compare clustered variables of groups. Bivariate analysis
(Student’s t-test) for independent samples was used for comparison of
normally distributed numerical variables. Normally distributed data were
expressed as mean ± standard deviation (SD) and compared by means of the
2-sample t test. Non-normally distributed data were expressed as median
with 95% confidence intervals and compared by means of the Mann-Whitney
U test. The Spearman rank correlation was used to examine the influence
of valve size on MES counts. The multivariate analysis was performed by
line regression method. P< 0.05 was considered as statistically
significant.
3. Results
In group A MES were detected in 62 % of the patients, in group B in 46%
of the patients (Fig.1). The difference between the two groups was not
statistically significant (p=0.2).
When studying the association between possible risk factors and MES we
found some difference between group A and group B.
In group A risk factors associated with the number of MES were smoking,
previous cardiovascular operations and EUROScore points (p<0.05) (Table
1).
In patients with EUROScore >5 MES were detected in 98% of the patients.
In the multivariate analysis all three risk factors remained
independently significant.
There was no correlation between the total number of MES and
anticoagulation (INR and anticoagulation therapy), atrial fibrillation,
cholesterol level, carotid artery disease, NYHA class, gender, age,
valve type or valve position.
There was a significant correlation between postoperative cerebral
events(stroke and TiA) and the number of MES in MES-positive group
(Fig.2).
In group B the following risk factors were associated with the number of
MES: age, uremia, atrial fibrillation and EUROScore.
In the multivariate analysis only two of the risk factors (atrial
fibrillation and uremia) remained independently significant. None of the
patients in group B experienced postoperative cerebral events.
4. Discussion
We were able to demonstrate MES in 62%of the valve patients and 46% of
the non-valve patients. The difference in MES between the two groups of
patients was not statistically significant. MES could be detected in all
types of cardiovascular patients. Patient related risk factors
associated with MES were different between valve and non-valve patients.
Our finding, demonstrating higher number of MES in patients previously
operated on, is new and there is to our knowledge no information in the
previous literature on the significance of smoking.
We did not find any correlation between the number of MES and the valve
position. This is in agreement with previous studies (18-21).
Only few studies have compared MES and various valve types (31). We did
not see any significant difference with regard to MES comparing
Carbomedics and On-X valves.
Contrary to a previous study from our own institution on the Carbomedics
valve we did not find any correlation between MES and valve size (18).
This discrepancy might be explained by the different types of equipment
and monitoring site used.
There is no data in the literature on the correlation between kidney
function and MES.
Our study confirms the findings of Kofidis at al.(20) showing no
correlation between MES and INR level.
There was no correlation between MES and NYHA class, diabetes,
hypercholesterol level, age and gender in the group of valve patients
(24-27). However, age was significantly important in non-valve patients.
In the agreement with Braekken et al.(22), we did not find atrial
fibrillation to be associated with MES in heart valve patients, however,
there were significantly more MES in non valve patients with atrial
fibrillation.
Theoretically, MES could have their origin in local plaques in the
carotid arteries, however, we did not find an increased number of MES in
our patients with known carotid artery disease.
There is no data in the literature on EUROScore and MES. We regard this
association to indicate that there are more MES in patients with more
advanced diseases.
In valve patients we found a significant association between the total
number of MES and postoperative stroke in MES-positive patients. This is
in agreement with previous reports (18-21). If it will b possible to
evaluate not only stroke or TIA history in valve patients but also
cognitive disorders, the correlation will be significant at the whole
valve patient group. The reason, we think, is that cognitive disorders
not always connecting by physicians with mechanical valve presence.
There is strong evidence that MES are really associated with bubbles or
solid microemboli (11,28,29,).
This is no agreement on the best way to monitor cerebral emboli. The
method used in the present study is simple to use, cheep and is able to
detect the highest number of MES due to one study(36).
A newly developed microemboli detector, EMEX-25, from Hatteland
Instrumentering, Norway was used for the measurements. This type of
investigation is new in valve patients, however, it has been used in
other types of patients (35,36).
It is of great importance to distinguish between real MES and artifacts.
The origin of artifacts could be multiple. The most important are
unintended movement of the probe, signals from movements of the vessel
wall and external noise sources such as diathermy. An emboli burst (MES
signal) would have a maximum possible duration time defined by the
transition time of the emboli through the sampling volume extension of
the probe and the minimum detectable velocity of the instrument.
Typically this maximum burst time would be 200 to 300 ms, the reason for
why some investigators defines a maximum burst duration time as a
criteria for valid MES signal detection. However this criteria is not
very sensitive because the noise sources mentioned above all could give
shorter bursts time comparable to those arising from true MES signals.
A more sensitive technique would be to analyze the behavior of the mean
velocity. Both the unintended movements of the probes as well as vessel
wall movement tended to give velocity components of very low speed and
with high energy compared to the background blood flow. These components
caused instability in the waveform of the mean velocity. Thus EMEX-25
was implemented with and algorithm analysing the waveform and stability
of the mean velocity. The algorithm was regarded to be superior to a
simple bursts length evaluation for removal of possible artifacts.
During the measurement, high attention was made to find a stabile signal
pattern with as minimum variation of the envelope signal amplitude as
possible. False positive detections or artifacts could occur when the
sampling point was close to the wall of the artery. Those spikes,
however, was recognized by typically being synchronized to the pulsation
of the flow, and was minimized to obtains as stabile signal as possible.
Another important detailed was to keep the probe axis in the sagital
plane to the trachea. By preventing the probe axis to cross the
air-tissue surface of trachea, any high energy disturbing reflections
from this region was avoided.
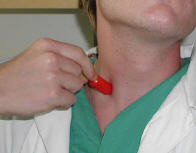
When a probe position giving stable signals was found, a recording
period of 2 times 2 minutes was applied in all patients and controls.
One can speculate whether our findings indicate a gaseous nature of the
MES rather than a solid.
Our method does not permit discrimination between gaseous MES and solid
MES.
Many authors suggest cavitations to be important (30,31). However, our
study clearly demonstrates that MES can be detected also in patients
without artificial heart valves.
|
|
Literature:
1. Koertke H, Minami K, Biaraktaris A, Wagner O, Koerfer
R. INR Self-management following Mechanical ytart valve replacement.
Journ of trombosis and trombolisis 2000; 9:41-45.
2. Svennevig JL. Off-pump vs on-pump surgery. A review. Scand Cardiovasc
J 2000;34:7-11.
3. Braekken S, Russell D, Brucher R, Abdelnoor M, Svennevig J. Cerebral
microembolic signals during cardiopulmonary bypass surgery. Stroke 1998;
28:1988-1992.
4. Brucher R, Russell D, Background and principles. In: Tegeler CH,
Babikian VL, Gomes CR, editors. Neurosonology. St. Louis: Mosby-Year
book 1996:231-234.
5. Gallager EG, Pearson DT. Ultrasonic identification of sources of
gaseous microemboli during open heart surgery. Thorax 1973; 28:295-305.
6. Hatteland K, Semb BKH. Gas bubble detection in fluid lines by means
of pulsed Doppler ultrasound. Scan J Thorac Cardiovasc Surg. 1985;
19:119-123.
7. Spencer MP, Lawrence GH, Thomas GI, Sauvage LR. The use of ultrasonic
in the determination of arterial aeroembolism during open-heart surgery.
Ann Thorac Surg 1969; 8:489-497.
8. Padayachee TS, Parsons S, Theobold R, Linley J, Gosling RG, Deverall
PB. The detection of the microemboli in the middle cerebral artery
during cardiopulmonary bypass. Ann Thorac Surg 1987; 44:298-302.
9. Padayachee TS, Parsons S, Theobold R, Gosling RG, Deverall PB. The
effect of arterial filtration on reduction of gaseous microemboli in the
middle cerebral artery during cardiopulmonary bypass. Ann Thorac Surg
1988; 45:647-649.
10. Pugsley W. The use of Doppler ultrasound in the assessment of
microemboli during cardiac surgery. Perfusion 1989; 4:115-122.
11. Russell D, Madden KPClarc WM, Sandset PM, Zivin JA. Detection
arterial microemboli using Doppler ultrasound in rabbits. Stroke 1991;
22:253-258.
12. Harrison MJG, Pugsley W, Newman S, Paschalis C, Klinger L, Treasure
T, Aspey B. detection of middle cerebral amboli during coronary artery
bypass surgery using transcranial Doppler sonography. Stroke 1990;
21:1512.
13. Stump DA, Newman SP. Embolus detection during cardiopulmonary
bypass, In: Tegeler CH, Babikian VL, Gomes CR, eds. Neurosonology.
St.Louis, Mo:Mosby-year book 1996:252-255.
14. Clark RE, Brillman J, Davis DA, Lovell MR, Price TRP, Magovern GJ.
Microemboli during coronary artery bypass grafting: genesis and effect
on outcome. J Thorac Cardiovasc Surg 1995; 109:249-258.
15. Braekken S, Russell D, Brucher R, Abdelnoor M, Svennevig J. Cerebral
microembolic signals during cardiopulmonary bypass surgery. Stroke 1997;
28:1988-1992.
16. Barbut D, HintonRB, Szatrowski TP, Hartman GS, Breufach M,
Williams-Russo P, Charlson ME, Gold JP. Cerebral emboli detected during
bypass surgery are associated with clamp removal. Stroke 1994;
25:2398-2402.
17. Baker AJ, Naser B, Benaroia M, Mazer CD. Cerebr al mikroemboli
during coronary artery bypass using different cardioplegia techniques.
Ann Thorac Surg 1995; 59:1187-1191.
18. D’Alfonso A, Milano AD, Codecasa R, De Carlo M, Nardi C, Orlandi G,
Paoli C, Murri L, Borrtolotti U. High-intensity transcranial Doppler
signals in patients wearing heart valve prostheses: a prospective study.
G Ital Cardiol 1999; 29:401-410.
19. Livense AM, Bekker SL, Dippel DW, Taams MA, Koudstaal PJ, Bogers AJ.
Intracranial high.intensity transient signals after homograft or
mechanic aortic valve replacement. J Cardiovasc Surg (Torino) 1998;
39:613-617.
20. Kofidis T, Fischer S, Rainer L, Mair H, Deckert M, Haberl R,
Haverich A, Reichart B. Clinical relevance of intracranil high intensity
transient signals in patients following prosthetic aortic valve
replacement. Eur J Cardiothorac Surg 2002; 21:22-26.
21. Georgiadis D, Braun S, Uhlmann F, Bernacca GM, Zierz S, Zerkowski
HR. Doppler microembolic signals in patients with two diferent types of
bileaflet valves. J Thorac Cardiovasc Surg 2001; 121:1101-6.
22. Brakken SK, Reinvang I, Russell D, Brucher R, Svennevig JL.
Association between intraoperative cerebral microembolic signals and
postoperative neuropsychological dificit: comparison between patients
with cardiac valve replacement and patients with coronary artery bypass
grafting. J Neurology 1998; 65:573-576.
23. Pugsley W, Klinger L, Paschalis C, et al. The impact of microemboli
during cardiopulmonary bypass on neuropsychological functioning. Stroke
1994; 25:1393-9.
24. Buhre W, Buhre K, Aleksic I, Zenker D, Sonntag H, Weyland A.
Doppler-sonographic evidence of cerebral microembolism originating from
biventricular assist device. Thorac Cardiovasc Surg 2000; 48:300-302.
25. Knapperts VA, Tegeler CH, Furberg CD, Wesley DJ, Stewart KP, Kitzman
DW. Carotide doppler high intensity transient signals in dilated
cardiomyopathy. Am Heart J 2000; 140:1-4.
26. Batista P, Oliveira V, Ferro JM. The detection of microembolic in
patients at risk of recurrent cardioembolic stroke: possible therapeutic
relevance. Cardiovasc Dis 1999; 9:314-319.
27. Eicke BM, Barth V, Kukovski B, Werner G, Paulus W. Cardiac
microembolism: prevalence and clinical outcome. JNeurol Sci 1996;
136:143-147.
28. Declunder G, Lecroart JL, Lapeyre D, Gregoric I, Rose H, Tames D,
Frasier OH. Effects of myocardial contractility on microemboli
production by mechanical heart valves in a bovine model. Tex Heart Inst
J 2000; 27:236-239.
29. Conger JL, Deklunder GM, Lecroart JL, LaPeyre DM, Gregoric I, Rose
H, Wieting DM, Clubb Jr F, Frazier OH. A bovine model for detecting high
intensity transient signals originating from mechanical heart valves.
ASAIO J 2000; 46:344-350.
30. Rambod E, Beisaie M, Shusser M, Milo S, Gharib M. A physical model
describing the mechanism for formation of gas mocrobubbles in patients
with mitral mechanical valves. Ann Biomed Ing 1999; 27:774-792.
31. Telman G, Kouperberg E, Sprecher E, Yarnitsky D. The nature of
microemboli in patients with artificial valves. J Neuroimaging 2002;
12:15-8.
32. Baumgartner RW, Frick A, Kremer C, Oechslin E, Russi E, Turina J,
Georgiadis D. Microembolic signals counts increase during hyperbaric
exposure in patients with prosthetic valves. J Thorac Cardiovasc Surg,
2001; 122:1142-6.
33. Perthel M, Hasenkam JM, Nygaard H, Kupper W, Laas J. Turbulence and
high intensity transient signals (HITS) as a parameter for optimum
orientation of mechanical heart valves. Zeitschrift fur Kardiologie
2001; 90:100-4.
34. Roques F, Nashef SAM, Michel P, Gauducheau E, De Vincentiis C,
Baudei E, Cortina J, David M, Faichney A, Gabrielle F, Gams E, Harjula
A, Jones MT, Salamon R, Thulin L. Risk factors and outcome in European
cardiac surgery: analysis of the EuroSCORE multinational database of
19030 patients. Eur J CardioThorac Surg 1999; 15:816-823.
35. D. Georgiadis, R. W. Baumgartner, R. Karatschai, A. Lindner, H. R.
Zerkowski. Further evidence of gaseous embolicmaterial in patients with
artificial heart valves. J Thorac Cardiovasc Surg 1998; 115:808-10.
36. Volker A. Knappertz, Charles H. Tegeler, Curt D. Furberg, Deborah J.
Wesley, Kathryn P. Stewart, Dalane W. Kitzman. Carotid Doppler
high-intensity transient signals in dilated cardiomyopathy. Am Heart J
2000;140:2-4.
|


